At the beginning of your research project
From now on, when conducting international biological research, every scientist has the obligation to figure out what international and national laws apply to sampling in the host country. To fulfill the obligations under the Nagoya Protocol, it is essential to check the ABS Clearing-House and get in touch with the National Focal Point and/or Competent National Authorities that are listed there. You will need to tell them what you are planning to do and hopefully they can guide you on which permits and approvals are necessary to carry out your research.
In addition to what the local authorities advise and require, you need to also consider what you will do with the biological material in the short- and long-term. The documents obtained in the provider country (PIC, MAT, MTA, IRCC) should be drafted with anticipation of the scientific purpose and goals of the project: what kind of research do you plan to do with it, whether you will do sequencing, or if you will deposit new isolates in a culture collection, etc. These objectives must be compatible with the permits and approvals and explicitly allowed by the MTA.
TIP: For deposits at the DSMZ (and other culture collection) it is essential that the MTA or other documentation clearly states that newly obtained microorganisms can be deposited in the public collection and distributed to users of the DSMZ. For many provider countries it is helpful to communicate to them that the DSMZ has a binding MTA of its own that limits usage of a DSMZ strain to non-commercial research use only and prohibits subsequent (i.e., third party) distribution of the strains. The International Code of Nomenclature for Bacteria requires deposit in two international collections and this should also be agreed to in advance in the MTA.
The DSMZ does NOT accept deposits if the host country requires that we obtain permission from them for future distribution of the strain. The administrative effort to obtain individual permissions from the country of origin is not only logistically unrealistic, but permission-blocked strains do not comply with the International Code of Nomenclature for Bacteria (Rule 30), which states that strains must be available “without restrictions” and, as such, these strains are scientifically irreproducible and do not meet standards of Good Scientific Practice.
As a registered collection, the DSMZ is required to provide all strains in our public collection with accompanying Nagoya Protocol documentation to our customers. The Nagoya Protocol has been in force since 2014 and new countries are joining all the time. Therefore, the legislative process in countries is at different stages; in some countries new laws are being written or existing ones are being revised.
If the DSMZ obtains new information about laws of the country of origin of the resource during the deposit process, the DSMZ must remove the resource from the catalog until the depositor has subsequently submitted the relevant documents. We are happy to assist you in obtaining the documents so that the genetic resource can be deposited and made available to science in compliance with the laws.
Ready to deposit at the DSMZ?
All DSMZ depositors of non-human biological material MUST provide the following information:
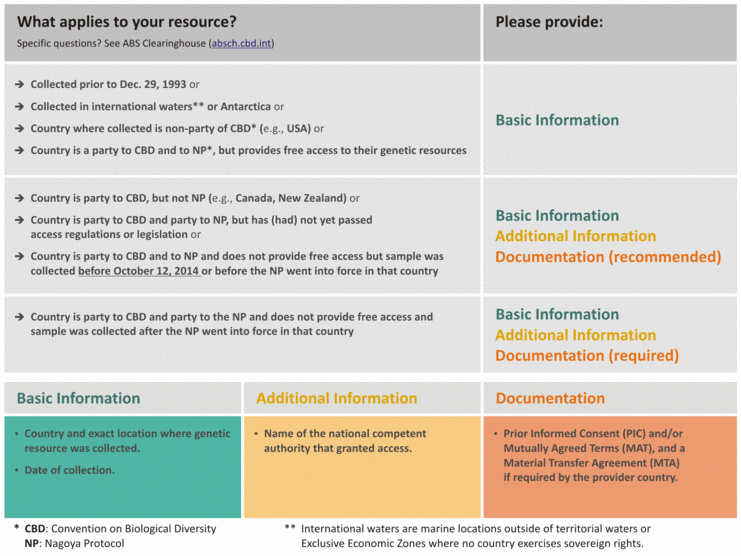
1) The country where the genetic resource was collected (not where it was isolated). (Samples collected in “International Waters” or in “Antarctica” should be listed under these locations.)
Note: Country includes the land, territorial waters, and Exclusive Economic Zone (which can extend up to 200 nautical miles offshore as defined by the United Nations Conference on the Law of the Sea), or any other area where the country exercises sovereign rights. This website helps you to determine, if a location belongs to international waters.
Some countries (for example Australia, Denmark, France, New Zealand, Netherlands, Norway, United Kingdom, United States) have external territories that are geographically separated from the governing country, for example overseas collectives of France, British Overseas Territories, dependent territories of the government of the United States, Australian external territories, and might have different regulations concerning access to genetic resources than the main land (e.g., Denmark’s ratification of the Nagoya Protocol does not apply to Greenland). It is especially important to check regulations in these areas in the ABS Clearing House and with the country’s National Focal Point.
2) The exact location within the country where the genetic resource was collected (GPS coordinates mandatory).
3) The date of collection (sampling).
4) CBD and Nagoya Protocol Check. Based on the information provided above, our online accession form for microbial deposits will automatically determine whether the genetic resource is regulated by the CBD or the NP.
An online accession form is currently only available for microbial deposits. For bacteriophage, plant virus, and animal cell line deposits, the PDF form should be completed using the ABS Clearinghouse and questions directed to the appropriate curator.
5) CBD and Nagoya Protocol Additional lnformation. If, based on the check described in 4 above, it is determined that your resource is regulated by the CBD or NP, you will be asked to provide the name of the national competent authority that granted access to the genetic resource and possibly to upload documentation showing Prior Informed Consent (PIC) and/or Mutually Agreed Terms (MAT), and a Material Transfer Agreement (MTA) from the provider country. These could be contained in a single document (such as an internationally recognized certificate of compliance (IRCC)) or multiple separate documents. If the field in the accession form has a red margin, the information is required. If the field is white, the information is recommended but not required.
6) During the deposit process, you will be asked whether all other laws in the country of origin were complied with and if any accompanying documentation was obtained (e.g., permits for export, local/indigenous authorities, national park, etc.)? These documents can then be uploaded.
Failure to provide the information above could result in rejection of a deposit at the DSMZ.
The following chart may be useful in determining what information to have ready during the deposit process: